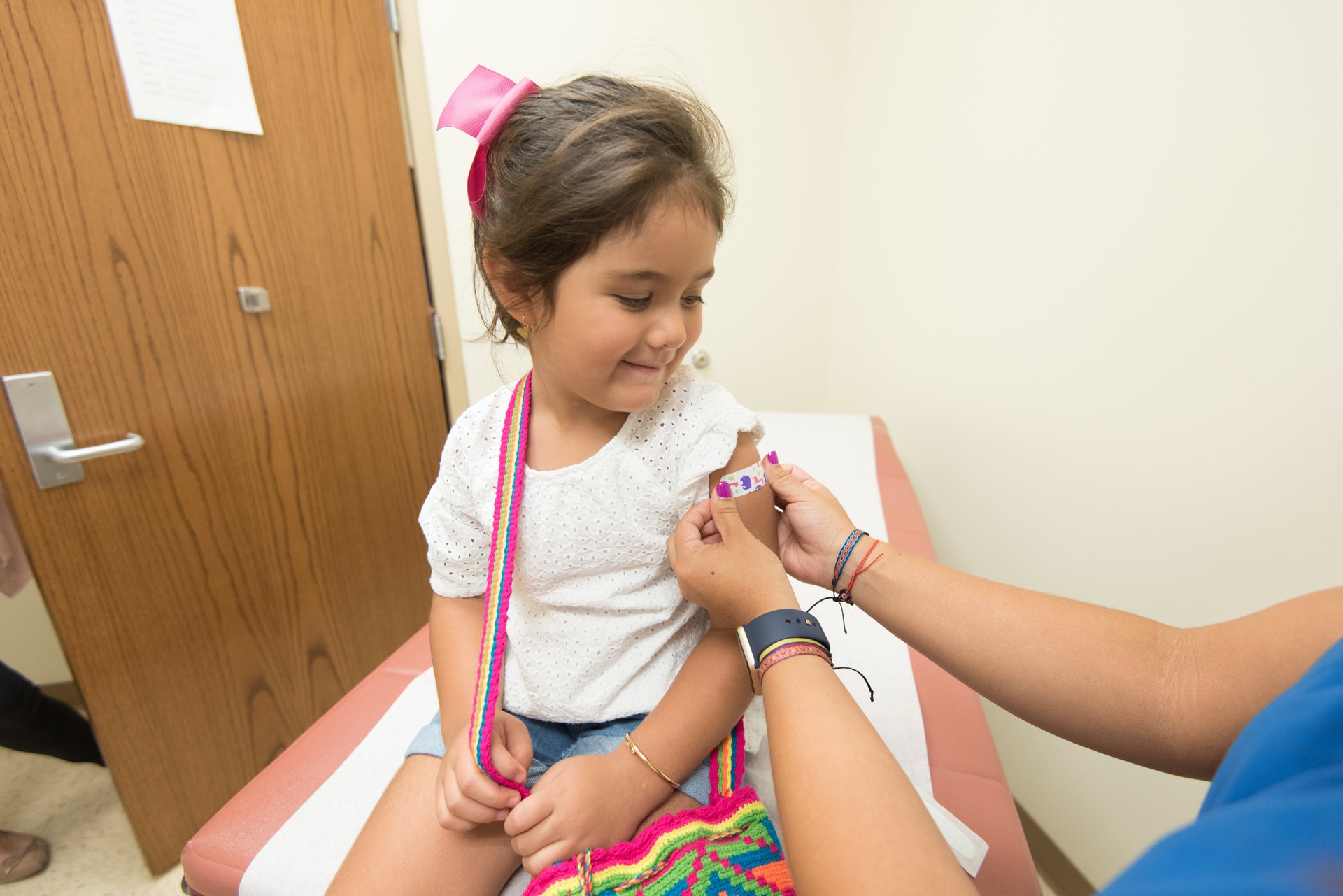

An estimated 6000 additional Putnam residents, five to eleven years of age, are now eligible for COVID shots and the Putnam County Department of Health has scheduled four clinics to vaccinate them, along with providing boosters for eligible adults. Afternoon clinics are scheduled for November 18 and 23, and December 9 and 14, and will run from 2 to 4:30 p.m. Children vaccinated at the first clinic in Carmel Plaza on November 18, can receive their second shot on December 9 in Carmel and be fully protected before Christmas Day. The clinics on November 23 and December 14 will be held at the Philipstown Recreation Center, with full protection by New Year's Eve. Online registration and further information will be shared on the health department's social media platforms and posted on their webpage at
https://www.putnamcountyny.com/health/covid19/#vaxinfo
The authorization of these youngsters comes as most clusters of COVID cases in Putnam County have affected schools and daycare centers where children gather. The most impacted school districts last month were Mahopac and Putnam Valley. The benefits for vaccinated children go beyond protection from infection. Fully vaccinated students of all ages, who remain asymptomatic, are not required to quarantine if exposed to a classmate or other person with COVID, allowing them to continue attending school and other activities.
We are working closely with local pediatricians in this phase of vaccinations, explained Kathleen Percacciolo, RN, supervising public health nurse. We are offering these Pfizer vaccination opportunities to assist them in their campaign to vaccinate this 5- to 11-year-old age group. Putnam pediatricians have been and continue to be the lead vaccine providers for children. One day COVID vaccination will become as routine as other childhood immunizations.
Now, these young children have the chance to join the more than 73,000 residents who have already received their first doses. In addition to providing first shots for these children, the PCDOH will offer booster shots and third doses to eligible adults at the same clinics. Eligibility depends on which shot was initially received. Anyone who received Moderna or Pfizer is eligible for a booster if they are 65 years and older, or 18 years and older and have certain health conditions, or live or work in a high-risk setting. These individuals must wait at least six months after their second dose. Those recipients who are 12 years and older, with weakened immune systems, (for example from cancer treatments) are eligible for a third dose if it is at least two months from their last shot. If it has been over two months, they are eligible for a booster. Booster and third doses are technically different and may vary clinically in the dosage. For individuals who received the Johnson & Johnson shot, and are 18 years of age and older, should get a booster if at least two months has passed since their shot.
The mission of the Putnam County Department of Health, nationally accredited by the Public Health Accreditation Board (PHAB), is to improve and protect the health of the Putnam County community, composed of nearly 100,000 residents. Core services include community health assessment, disease surveillance and control, emergency preparedness, environmental health protection, family health promotion and health education. For more information, please visit the Putnam County website at www.putnamcountyny.com, or visit the health department social media sites on Facebook, Twitter and Instagram.
For further information, contact: Shanna Siegel, Public Information Officer, at 845-656-4905.